Early Career Investigator Resources

The National Institutes of Health (NIH) Office of AIDS Research (OAR) coordinates HIV/AIDS research across NIH. The NIH provides the largest public investment in HIV/AIDS research globally. NIH OAR supports and promotes diversity to advance research and enhance innovation to end the HIV pandemic. As stated in the NIH Strategic Plan for HIV and HIV-Related Research, Goal 4 is to “build human resource and infrastructure capacity to enhance sustainability of HIV research discovery and the implementation of findings by a diverse and multidisciplinary workforce.”
To advance these goals, NIH OAR gathered input from a broad range of audiences on how to improve outreach to the next generation of HIV investigators and effectively address training and capacity-building programs. (Learn about select community engagements in this recent summary of OAR-hosted listening sessions.) In response to the significant input received, NIH OAR developed an HIV early career investigator page to centralize relevant information for investigators and provide easy access to grant opportunities, basic information, and other resources, such as training and capacity building programs.
NIH OAR 2025 Workshop for Early Career Investigators in HIV
The NIH Office of AIDS Research, along with partners across NIH, will hold the fourth annual virtual workshop for students, postdocs, and new HIV investigators on Friday, September 26, 2025, from 11 a.m. to 3 p.m. ET. Register and learn more on the event webpage.
Learn From Past ECI Events
NIH OAR 2024 Workshop for Early Career Investigators in HIV: Learn about the third annual virtual workshop on the event webpage, read the Executive Summary, and watch video recordings of the workshop.
NIH OAR 2023 Workshop for Early Career Investigators in HIV: View mentor presentations from the 2023 workshop at the links below.
Mallory Johnson, Ph.D., University of California, San Francisco
Always Have a Plan But Always Be Ready to Not Follow It
Carlos Rodriguez-Diaz, Ph.D., M.P.H.E., MCHES, George Washington University
Conducting Community-Centered HIV Research With Latinx Populations
David Chang, Ph.D., currently OAR Senior Science Advisor; previously NIH Center for Scientific Review
The NIH Grant Application Cycle: From Submission to Notice of Award
(Note: Some information may be out of date as there have been changes in the NIH review process.)
- NIH OAR Data Hub
- Explore the NIH HIV research portfolio with a new tool that allows users to search and filter awarded projects by research topic, funding opportunity, organization, and more.
- NIH HIV/AIDS Research Priorities and Guidelines for Determining HIV/AIDS Funding
- Find information from NIH OAR about NIH HIV/AIDS research priorities and guidelines for allocation of HIV funding for fiscal years 2021-2025.
- NIH Grants and Funding
- Find application instructions and guidance to submit grant applications to the NIH, the Centers for Disease Control and Prevention, the Food and Drug Administration, and the Agency for Healthcare Research and Quality.
- Listen to the NIH All About Grants Podcasts to hear about the ins and outs of NIH funding and get insights on grant topics.
- NIH Research Training and Career Development
- Explore individual and institutional NIH research training and career development (K award) opportunities at the undergraduate, graduate, and postdoctoral levels.
- Save the date for the 2025 Workshop for Early Career Investigators in HIV on September 26, 2025.
Use these dates and resources to guide your grant submission plans.
- Standard Due Dates
- HIV Dates
- Cycle I Due Date– May 7
- Cycle II Due Date – September 7
- Cycle III Due Date – January 7
- Review and Award Cycles (scroll down the page)
- Center for Scientific Review (CSR) Resources and Opportunities
- HIV/AIDS Study Sections at CSR
- Quickly find the most appropriate study section.
- CSR Assisted Referral Tool (ART)
- Use the tool to match your abstract or specific aims to a study section/scientific review group.
- Early Career Reviewer (ECR) Program
- Peer Review Webinars and Videos
- HIV/AIDS Study Sections at CSR
Visit the NIH ICO websites to learn about HIV research and funding opportunities, such as the following –
- Fogarty International Center (FIC)
- National Cancer Institute (NCI)
- National Heart, Lung, and Blood Institute (NHLBI)
- National Institute on Aging (NIA)
- National Institute of Allergy and Infectious Diseases (NIAID)
- Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD)
- National Institute of Dental and Craniofacial Research (NIDCR)
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
- National Institute on Drug Abuse (NIDA)
- National Institute of Mental Health (NIMH)
- National Institute of Neurological Disorders and Stroke (NINDS)
- National Library of Medicine (NLM)
- Office of Research on Women’s Health (ORWH)
- ORWH is offering Science Policy Scholar Travel Awards to support the development of junior investigators who are interested in women’s health or sex and gender differences and are also interested in research policy. Applications are due by February 1, 2023.
- Building Interdisciplinary Research Careers in Women's Health (BIRCWH)
- E-Learning
- Sex as a Biological Variable (SABV) Policy
- Understudied, Underrepresented, and Underreported (U3) Research
- Bench to Bedside: Integrating Sex and Gender to Improve Human Health Course
Learn more about the programs, conferences, and opportunities offered by NIH.
- AIDS Research Centers Program
- Avant-Garde Award Program for HIV/AIDS and Substance Use Disorder Research
- Avenir Awards for HIV/AIDS Research (Exclusive to early career investigators)
- Centers for AIDS Research (CFAR)
- Centers for AIDS Research Sites: CFARs located throughout the United States.
- Centers for AIDS Research (CFAR) Site Contacts: Links to individual sites.
- Early-Stage Investigator HIV/AIDS Research Using Nonhuman Primate (NHP) Models (R21 Clinical Trial Not Allowed)
- Early-Stage Investigator Research Using Nonhuman Primate (NHP) Models (R21 Clinical Trial Not Allowed)
- HEAL Initiative: The Helping to End Addiction Long-term
- HIV/AIDS Scholars Using Nonhuman Primate (NHP) Models Program (K01 Independent Clinical Trial Not Allowed)
- IMPAACT ECI program, including the award notification, application, and budget-related documents
- Mentored Career Development Program (K01) for Early Stage Investigators Using Nonhuman Primate Research Models (K01 Independent Clinical Trial Not Allowed)
- Mentored Research Scientist Development Award (Parent K01 - Independent Clinical Trial Not Allowed)
- NIAID New Innovators Awards (DP2 Clinical Trial Not Allowed)
- Questions and Answers for PAR-20-259, NIAID New Innovators Awards (DP2 Clinical Trial Not Allowed)
- Research Centers in Minority Institutions Program
- Stephen I. Katz Early Stage Investigator Research Project Grant (R01 Clinical Trial Not Allowed)
The HIV Vaccine Trials Network (HVTN) is the world’s largest publicly funded multi-disciplinary international collaboration facilitating the development of vaccines to prevent HIV/AIDS. The HVTN conducts all phases of clinical trials, from evaluating experimental vaccines for safety and immunogenicity to testing vaccine efficacy. Explore HVTN to learn about opportunities and programs for new investigators such as the Scholar Awards.
- Scholar Awards: The Research and Mentorship Program (RAMP) aims to recruit and retain a new generation of HIV vaccine researchers by providing structured mentorship and project funding to African American/Black and Latinx U.S. medical students.
- Submit an Idea or Proposal: HVTN welcomes the ideas of the scientific community regarding new proposals for HIV vaccine studies that are not currently being planned or pursued.
- Ancillary/Exploratory Studies: The HVTN shares data and specimens collected in its studies with investigators who would like to use HVTN’s materials to answer research questions related to vaccinology, immunology, or HIV/AIDS.
These visuals outline career path options and examples of NIH-funded mechanisms available to individuals in different stages of their careers. There are a variety of paths to pursue. For more information about individual funding mechanisms, see NIH Research Training and Career Development Programs.
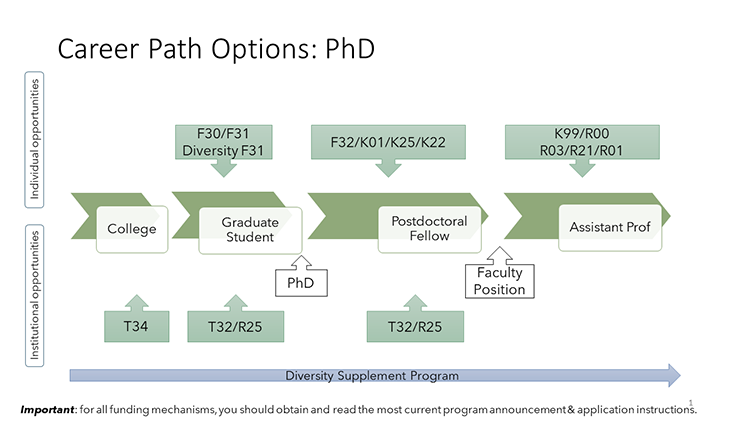
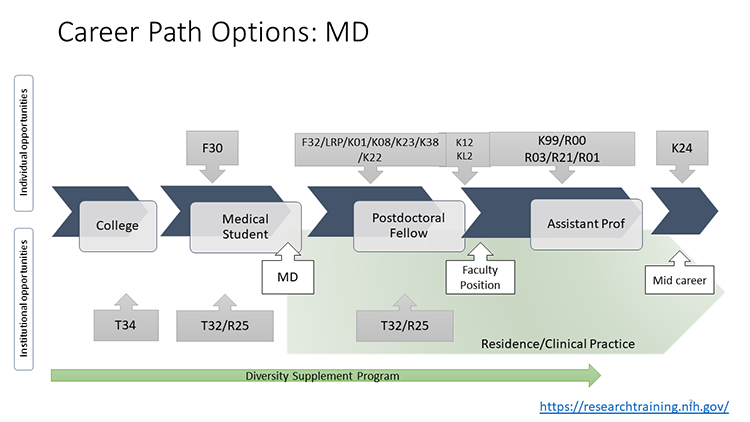
HIV Funding Opportunities
This list of Funding Opportunity Announcements (FOAs) may include HIV/AIDS receipt dates. Please note that some FOAs allow HIV/AIDS applications but they may have specific requests such as all AIMS should be related to HIV research. Contact the Program Officer indicated in the individual funding opportunity to request further details.
This page last reviewed on November 19, 2025


